The DNA Damage Response (DDR)
- Protects healthy cells from DNA damage
- Prevents DNA damage from accumulating to toxic levels
- Is required for normal cell survival and prevention of cancer-causing mutations

Targeting the Totality of the DDR in Cancer
- HRD tumors
- PARPi resistance
- Small cell lung cancer
- Combination therapies
- 10 to 15% of all cancers
- High prevalence in sarcomas
- Activation of cGAS/STING
- IO combination
Inhibitors of the DDR
Tumor cells require DNA mutations for disease progression and switch off DDR components to promote cancer development. However, this makes cancer cells reliant on remaining DDR pathways to keep DNA damage below the toxic levels that trigger cell death.
This delicate balance makes tumor cells much more vulnerable than normal cells to inhibitors of DDR proteins. In this way, by targeting selective DDR processes, our technology aims to kill cancerous cells and treat malignant disease while leaving healthy cells intact.
Unmet Need
Despite significant advances in oncology therapeutics, DNA-damaging modalities, such as chemotherapy and radiation therapy continue to be the most used treatment strategies across a wide range of cancer types. Unfortunately, these treatments come with limitations and a substantial proportion of cancers either fail to respond or acquire resistance over time.
Current DNA-damaging therapies are toxic to normal tissue
An inability to distinguish between cancerous and normal tissue yields toxicity, limits ability to dose, and compromises efficacy
Resistance to current anticancer agents, including PARP inhibitors
Up to 50% of patients with BRCA-deficient tumors fail to respond to PARP inhibition, the standard of care in many advanced cancers. Those who initially respond will eventually develop acquired resistance and the treatment will ultimately fail
Dependence on DDR-driven processes
In approximately 10% to 15% of all tumors, telomeres—specialized structures that protect chromosome ends from progressive degradation—are extended through the DDR-driven ALT pathway and are able to achieve replicative immortality. Many ALT-positive tumors are associated with poor prognosis and have a lack of targeted therapies
Limited ability to reach the immune system
A substantial portion of malignant disease is not amenable to immunotherapy, a strategy that has been proven to be highly effective in treating select cancers
Our portfolio exploits the totality of the DDR to address cancers with significant unmet needs.
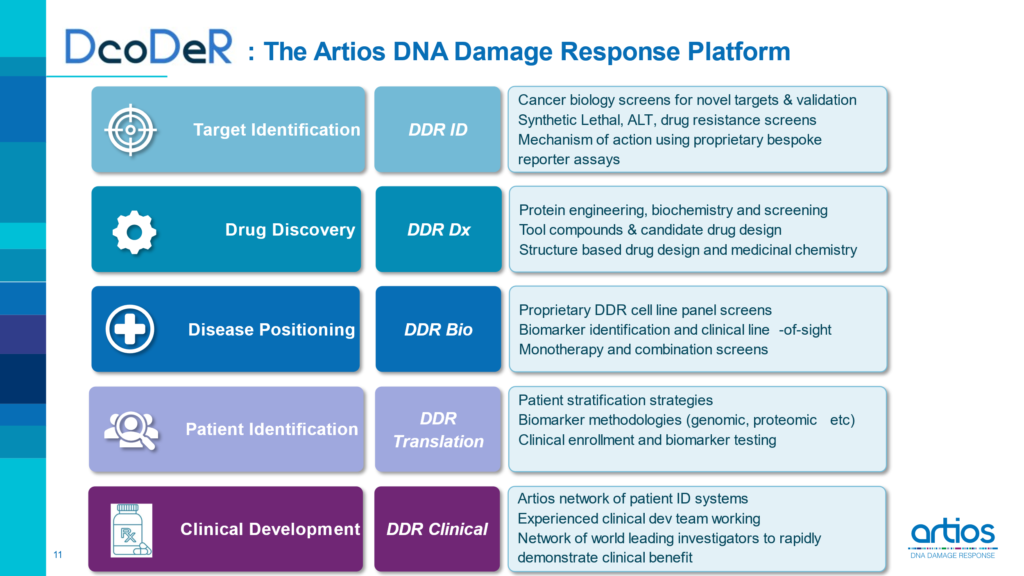
PipelineDcoDeR: A World Class DNA Damage Response Discovery Platform